|
|
|
|
The website to the left shows how to perform various Equilibrium calculations. Read this webpage then do the problems in the PDF on the right.
|
| |||||||
Le Châtelier's Principle
Le Châtelier's principle states that if a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium shifts to counteract the change to reestablish an equilibrium. If a chemical reaction is at equilibrium and experiences a change in pressure, temperature, or concentration of products or reactants, the equilibrium shifts in the opposite direction to offset the change.
Le Châtelier's principle states that if a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium shifts to counteract the change to reestablish an equilibrium. If a chemical reaction is at equilibrium and experiences a change in pressure, temperature, or concentration of products or reactants, the equilibrium shifts in the opposite direction to offset the change.
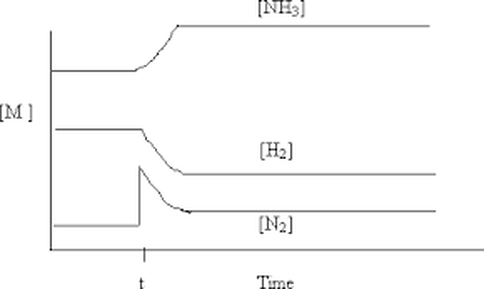
Look at the graph above and explain what has happened at t and what this does to i) Kc
ii) the concentration of each molecules in the reaction.
ii) the concentration of each molecules in the reaction.
|
| ||||||||||||||||||