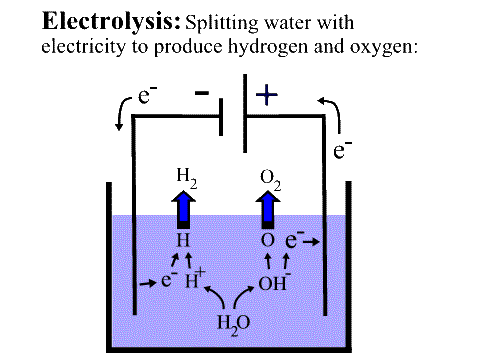
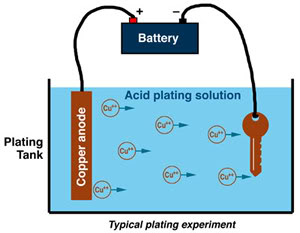
ELECTROCHEMISTRY AND CORROSION
|
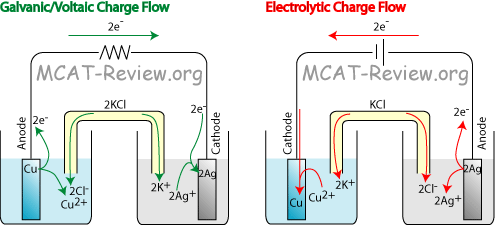
All About Electrochemistry gives a comprehensive description of electrochemistry including a history of the science. Chapters 2,3,6 and 7 are the most important.
|
|
Watch the video on the right to help understand the assigning of oxidation numbers, once you have this understanding we can now start to apply the method of assigning oxidation numbers to help us in looking at ANY chemical equation and determine if a redox reaction has occurred. To understand how watch the clip directly below called Redox reactions with Mr Anderson at Bozeman Science up until about 8:30.
|
|
|
|
|
|
|
|
|
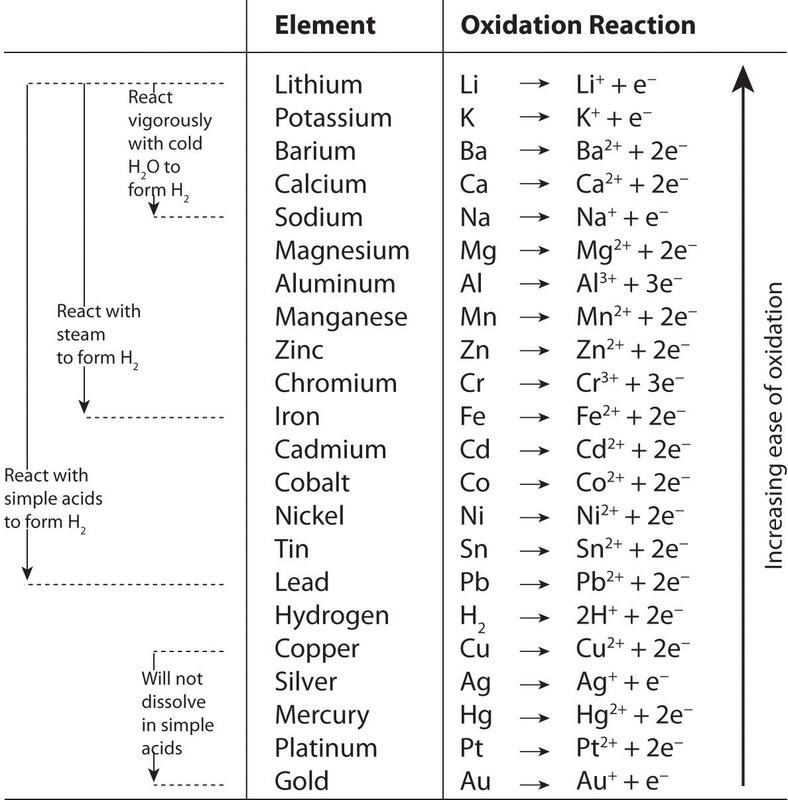
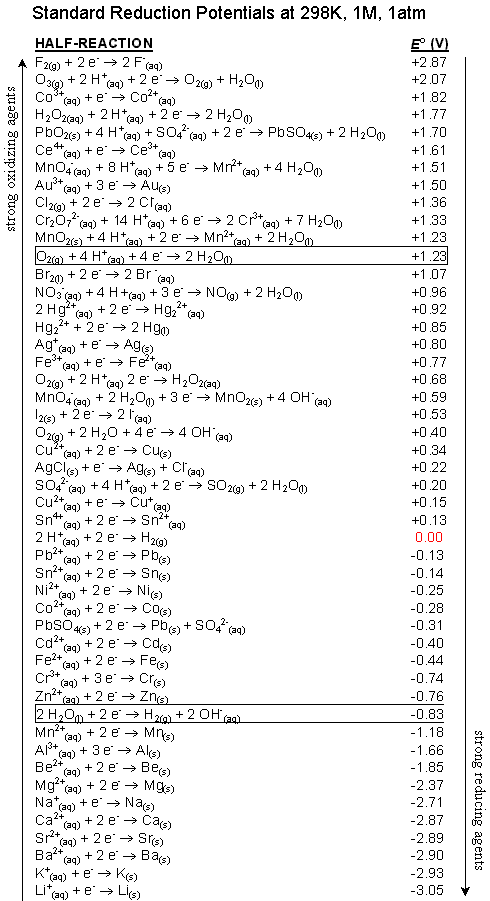
Rules for balancing redox reactions in acidic solution
1.Assign oxidation numbers to elements 2.Write the 2 half-reactions: reductionn and oxidation 3.Balance all elements except O and H 4.Balance O using H2O and balance H using H+ (on the other side, as many as needed to make H2O molecules) 5.Balance the charges by adding electrons 6.Multiply half-reactions by factors that cancel out electrons 7.Add half-reactions back together |
Points to remember:
1) Electrons NEVER appear in a correct, final answer. In order to get the electrons in each half-reaction equal, one or both of the balanced half-reactions will be multiplied by a factor.2) Duplicate items are always removed. These items are usually the electrons, water and hydrogen ion
1) Electrons NEVER appear in a correct, final answer. In order to get the electrons in each half-reaction equal, one or both of the balanced half-reactions will be multiplied by a factor.2) Duplicate items are always removed. These items are usually the electrons, water and hydrogen ion
| redox_reactions_worksheets.doc | |
| File Size: | 34 kb |
| File Type: | doc |
|
|
|
|
To learn more about electrochemistry, visit this page
|
| ||||||||