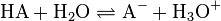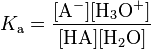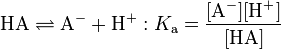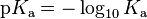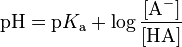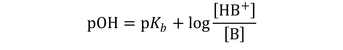Acid dissociation constant Ka |
An acid dissociation constant, Ka, (also known as acidity constant, or acid-ionization constant) is a quantitative measure of the strength of an acid in solution.
|
In aqueous solution, the equilibrium of acid dissociation can be written symbolically as:
where HA is an acid that dissociates into its conjugate base A−, and a hydrogen ion H+ which combines with a water molecule to make an hydronium ion H3O+.
The chemical species HA, A− and H3O+ are said to be in equilibrium when their concentrations do not change with the passing of time. The dissociation constant Ka is usually written as a quotient of the equilibrium concentrations (in mol/L), denoted by [HA], [A−] and [H3O+].
The chemical species HA, A− and H3O+ are said to be in equilibrium when their concentrations do not change with the passing of time. The dissociation constant Ka is usually written as a quotient of the equilibrium concentrations (in mol/L), denoted by [HA], [A−] and [H3O+].
In most cases (except the most concentrated aqueous solutions of an acid), the concentration of water does not singificantly as it reacts with the acid. It can therefore be taken as constant and be ignored. The reaction and the definition can then be written more simply
For many practical purposes it is more convenient to discuss the logarithmic constant, pKa
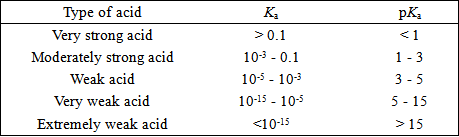
Relationship between Ka, pKa and acid strength:
The smaller the value of Ka, the larger the value of pKa, the weaker the acid.
The smaller the value of Ka, the larger the value of pKa, the weaker the acid.
Relationship between pH and pKa for a monoprotic acid:
If the pH of a solution of a weak acid and the pKa are known, the ratio of the concentration of the conjugate base to the concentration of the acid may be calculated.
Similarly for bases:
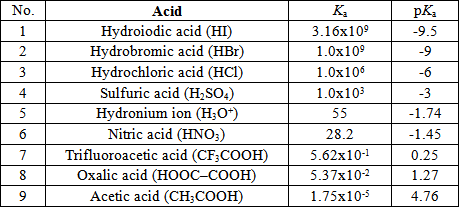
Links to table of Ka and pKa values
|
For a more comprehensive list of pKa and Ka values, click on the link on the right.
(Source: http://clas.sa.ucsb.edu/staff/Resource%20Folder/Chem109ABC/Acid,%20Base%20Strengt h/Table%20of%20Acids%20w%20Kas%20and%20pKas.pdf) |