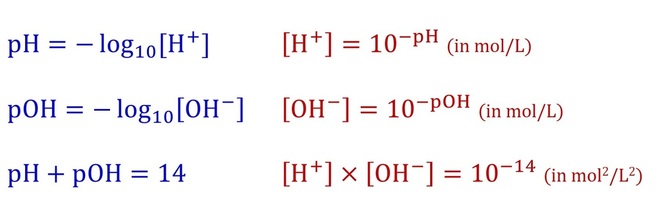Main relationships between pH, pOH, [H+] and [OH-] at 25◦C
|
The table on the right lists values of the ionic product of water Kw at different temperature and the corresponding pH for pure water. While the value of Kw is constant at a given temperature, it increases when temperature increases.
Note that since [H+]=[OH-] in pure water, the pH of pure water varies with temperature. |
|
|
Read the webpage on Kw to the left then complete worksheet 1 below.
|
Worksheets on pH of solutions. Some with answers:
|
Worksheet on molarity calculations. With answers:
|
More pH related calculations:
| ||||||||