Organic molecules and hydrocarbons
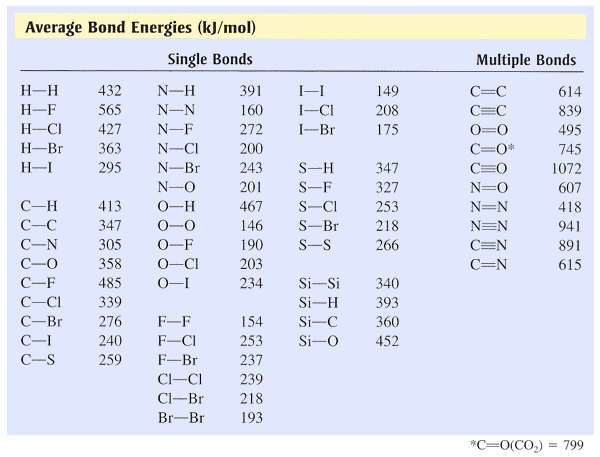
Bond energiesFor any particular chemical bond, the amount of energy to break the bond is the same as the amount of energy released when the bond is formed. The amount of energy required to break the bond is called bond dissociation energy. It is expressed in kilojoules per mole of bond broken or kJ/mol.
Each value of bond energy in the table on the right is the average enthalpy change to break all the similar bonds of a compound in the gaseous state. For instance, 435 kJ is required to break a single C-H bond in methane, but breaking all four bonds requires 1662 kJ. Thus, the average bond energy for a bond C-H in methane is 416 kJ/mol. |