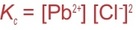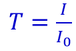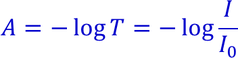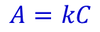WATER ANALYSIS
Under this page, you will find information regarding the unique physical and chemical properties of water, owing to the structure and composition of its constitutive molecule H2O, which is also discussed.
The second part provides background information on analytical techniques used to assess the quality of water.
The second part provides background information on analytical techniques used to assess the quality of water.
Solubility product constant Ksp
Solubility product constants are used to describe saturated solutions of ionic compounds of relatively low solubility. As seen in Term 3, a saturated solution of a salt is in a state of dynamic equilibrium between the dissociated ionic compounds in aqueous state (dissolved in water) and the undissolved solid.
Let's consider the equilibrium between the slightly soluble PbCl2 salt (lead(II) chloride) and its constitutive ions in aqueous solution.
Let's consider the equilibrium between the slightly soluble PbCl2 salt (lead(II) chloride) and its constitutive ions in aqueous solution.
In the case of salts, the constant of equilibrium Kc is called solubility product constant Ksp.
Starting from a sample of pure PbCl2 dissolved in water, the following relationship between the concentrations of Pb2+ and Cl- will be found at equilibrium: [Cl-]=2[Pb2+] or [Pb2+]=0.5[Cl-].
Substituting either relationship in the expression of Ksp, it comes Ksp=4[Pb2+]^3 or Ksp=0.5[Cl-]^3
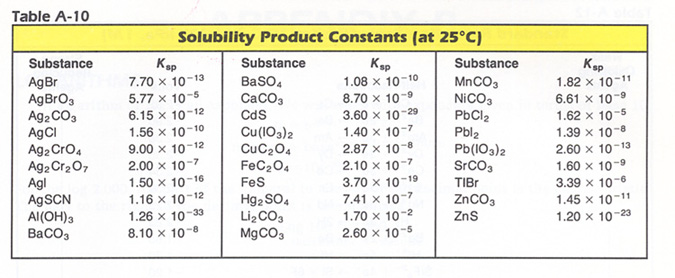
Therefore, the knowledge of Ksp of PbCl2 enables to predict the concentration of both ions in a saturated solution of the salt at a given temperature. It also tells whether a salt is very soluble, soluble, slightly soluble or insoluble. The smaller the Ksp value of a salt, the less soluble it is. Only Ksp for slightly soluble to very insoluble salt are generally reported in solubility product constants values. Examples of Ksp values at 25oC are shown in the table below.
(Remember that the only factor that affect the value of a constant of equilibrium, including Ksp, is the temperature.)
Starting from a sample of pure PbCl2 dissolved in water, the following relationship between the concentrations of Pb2+ and Cl- will be found at equilibrium: [Cl-]=2[Pb2+] or [Pb2+]=0.5[Cl-].
Substituting either relationship in the expression of Ksp, it comes Ksp=4[Pb2+]^3 or Ksp=0.5[Cl-]^3
Therefore, the knowledge of Ksp of PbCl2 enables to predict the concentration of both ions in a saturated solution of the salt at a given temperature. It also tells whether a salt is very soluble, soluble, slightly soluble or insoluble. The smaller the Ksp value of a salt, the less soluble it is. Only Ksp for slightly soluble to very insoluble salt are generally reported in solubility product constants values. Examples of Ksp values at 25oC are shown in the table below.
(Remember that the only factor that affect the value of a constant of equilibrium, including Ksp, is the temperature.)
UV-visible spectrophotometry
|
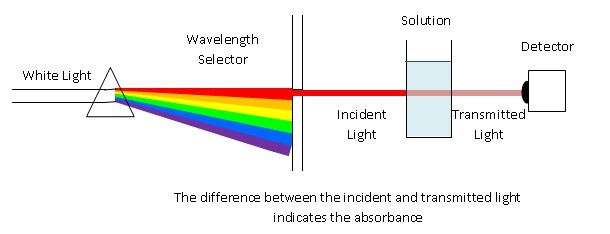
A spectrophotometer is employed to measure the amount of light that a sample of a pure substance in solution in a solvent absorbs.
The instrument operates by passing a beam of light through a sample and measuring the intensity of light reaching a detector. When a photon encounters an analyte molecule (the analyte is the molecule being studied), there is a chance the analyte will absorb the photon. This absorption reduces the number of photons in the beam of light, thereby reducing the intensity of the light beam. |
Used for the determination of phosphate and nitrate ions concentration in water.
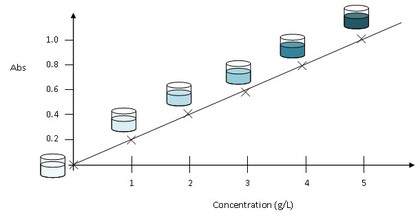
Calcuations associated with spectrophotometry
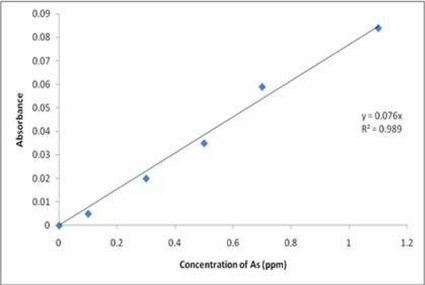
A practical example is shown below for the concentration of elemental arsenic in water. The absorbance values of standard solutions of arsenic were measured. The equation of the calibration curve enables to determine an unknown concentration of arsenic in a water sample from measuring the absorbance of a sample of the solution.
| spectrophotometry.xlsx | |
| File Size: | 17 kb |
| File Type: | xlsx |