Bonding and intermolecular forces
There are 3 types of bonds:
In addition, a number of weaker forces explain how molecular species interact with each other. The knowledge of intermolecular forces is essential to understand the physical properties of molecular compounds, such as their melting and boiling points.
The series of clips below explain the nature of metallic, ionic and covalent bonding and explore the similarities and difference between these forces. In addition, some information on intermolecular forces is provided.
- Metallic: explain the bonding between metal atoms and most physical and chemical properties of metal
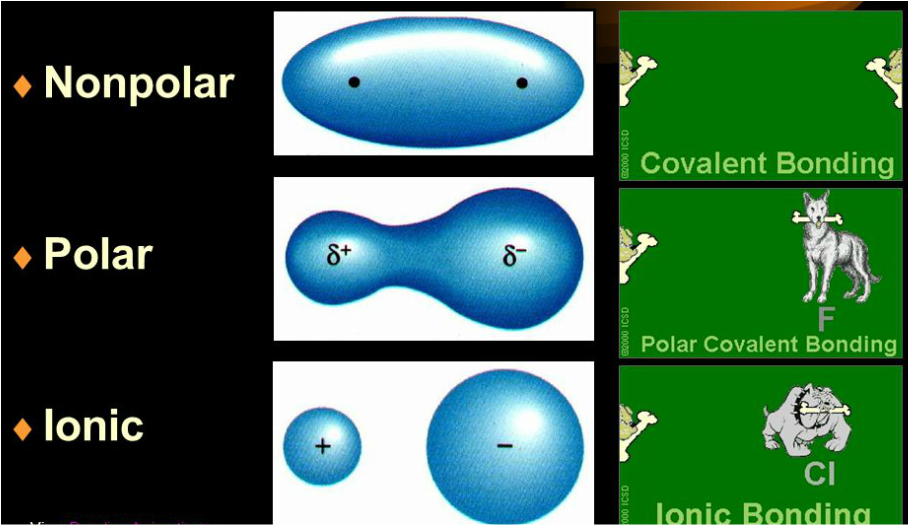
- Ionic: bond resulting from the reaction of a metal element with non-metal element where ions of opposite charge are produced. All salts and most minerals involve ionic bonds.
- Covalent: when 2 non-metal atoms (from the same element or from different elements) share electrons.
In addition, a number of weaker forces explain how molecular species interact with each other. The knowledge of intermolecular forces is essential to understand the physical properties of molecular compounds, such as their melting and boiling points.
The series of clips below explain the nature of metallic, ionic and covalent bonding and explore the similarities and difference between these forces. In addition, some information on intermolecular forces is provided.
Metallic bonding
Ionic and covalent bonding
Bonding Powerpoint
|
The Powerpoint on the right is a general explanation of electron configuration and then goes into the 3 types of bonding; ionic, covalent and metallic.
|
| ||||||
|
| |||||||||||||
Bond polarity
Intermolecular forces: an overview
|
A number of weaker forces explain how molecular species interact with each other. The knowledge of intermolecular forces is essential to understand the physical properties of molecular compounds, such as their melting and boiling points.
Understanding how intermolecular forces work require a good understanding of covalent bonding. While intermolecular forces are much weaker than metallic, covalent and ionic bonds, hydrogen bonding is the strongest form of intermolecular forces. |
| ||||||||||||